Medium Capsule Filters

Let's talk.
Send a message to our
industry experts.
Medium Capsule
Let's talk.
Send a message to our
industry experts.
Medium Capsule
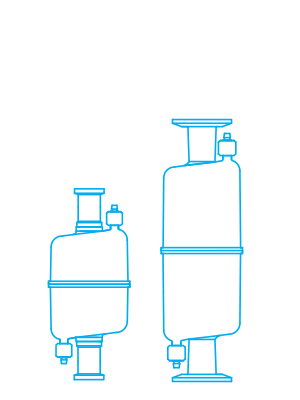
Meissner’s medium capsule filter (CF model) is available with a variety of inlet/outlet connections (hose barb, MNPT, sanitary or TC flange) for maximum flexibility and convenience. The capsule filter provides a nominal filtration area of 0.70 ft.2 (6.50 m2). Available with Meissner’s full range of sterilizing-grade filtration, clarification, purification, and prefiltration media with absolute removal ratings from 0.04 µm to 99 µm.
The CF capsule filter is available in gamma-irradiated models. The filters comply with European Commission Regulation No. 10/2011. Capsule filter materials of construction meet FDA standards for food contact per 21 CFR 177.
Filter Media Options
Membrane Filters for Liquid (Hydrophilic)
Microfiber Media Filters
Membrane Filters for Air/Gas (Hydrophobic)
Additional Filter Configurations
Frequently Asked Questions
What are Medium Capsule Filters?
Medium Capsule Filters are filtration devices designed for biopharmaceutical manufacturing processes. They consist of a filter membrane enclosed within a capsule housing, facilitating sterile filtration of liquids. These filters are available with various inlet and outlet connection types, including hose barb, MNPT, sanitary, or TC flange, to accommodate different system configurations.
What applications are Medium Capsule Filters used for?
Medium Capsule Filters are used primarily for sterile filtration, clarification, and particle removal in biopharmaceutical manufacturing. They are suitable for applications such as cell culture media filtration, buffer filtration, and final product sterilization where maintaining product integrity and sterility is critical.
What specifications or sizes are available for Medium Capsule Filters?
Medium Capsule Filters are offered with multiple inlet and outlet connection options, including hose barb, MNPT, sanitary, and TC flange fittings. Specific membrane materials, pore sizes, and surface areas depend on the filtration requirements but are selected to meet standard bioprocessing needs. Detailed size and specification data should be obtained from product datasheets or technical support.
What makes Medium Capsule Filters unique?
Medium Capsule Filters provide versatility through a variety of connection types, enabling easy integration into diverse bioprocess systems. Their capsule design ensures a closed, sterile environment during filtration, reducing contamination risk. The availability of multiple inlet/outlet configurations supports compatibility with different tubing and piping standards used in biopharmaceutical manufacturing.
How do Medium Capsule Filters integrate with other systems?
Medium Capsule Filters integrate with upstream and downstream bioprocess equipment via their standardized inlet and outlet connections. They can be connected directly to tubing or piping systems using hose barb, MNPT, sanitary, or TC flange fittings. This flexibility facilitates seamless incorporation into single-use or stainless-steel process lines.
Are Medium Capsule Filters suitable for regulated biopharmaceutical manufacturing?
Yes, Medium Capsule Filters are designed to meet the stringent requirements of regulated biopharmaceutical manufacturing. Their construction materials and design support compliance with industry standards for sterility, biocompatibility, and extractables/leachables. Proper use and validation ensure they are suitable for GMP environments.
Can Medium Capsule Filters be customized?
Customization options for Medium Capsule Filters may include selection of membrane material, pore size, and connection types to meet specific process requirements. Custom configurations can be provided to align with unique system designs or filtration needs, subject to technical feasibility and manufacturing capabilities.
What other products are commonly used with Medium Capsule Filters?
Medium Capsule Filters are commonly used alongside upstream components such as UltraClamp™ Sanitary Clamps, Universal Lab Stand, and Stainless Steel Stand for FlexFill® Biocontainers. Downstream, they are often paired with process monitoring devices such as the SGT (Single-Use Gauge Tee). These complementary products support fluid handling, system support, and process control in biopharmaceutical workflows.